
Top Neurology CRO – Contract Research Organization
If you’re a physician looking to fund a clinical trial, you may be wondering which CRO is right for you. This article will discuss the terms Vial Contract Research Organization (CRO), Neurological Clinical Research Institute (NCRI), Cognitive Clinical Trials (CCT), and QPS. It will also explain why these Top Neurology CROs are most important to the process of conducting clinical trials.
Vial Contract Research Organization (CRO)
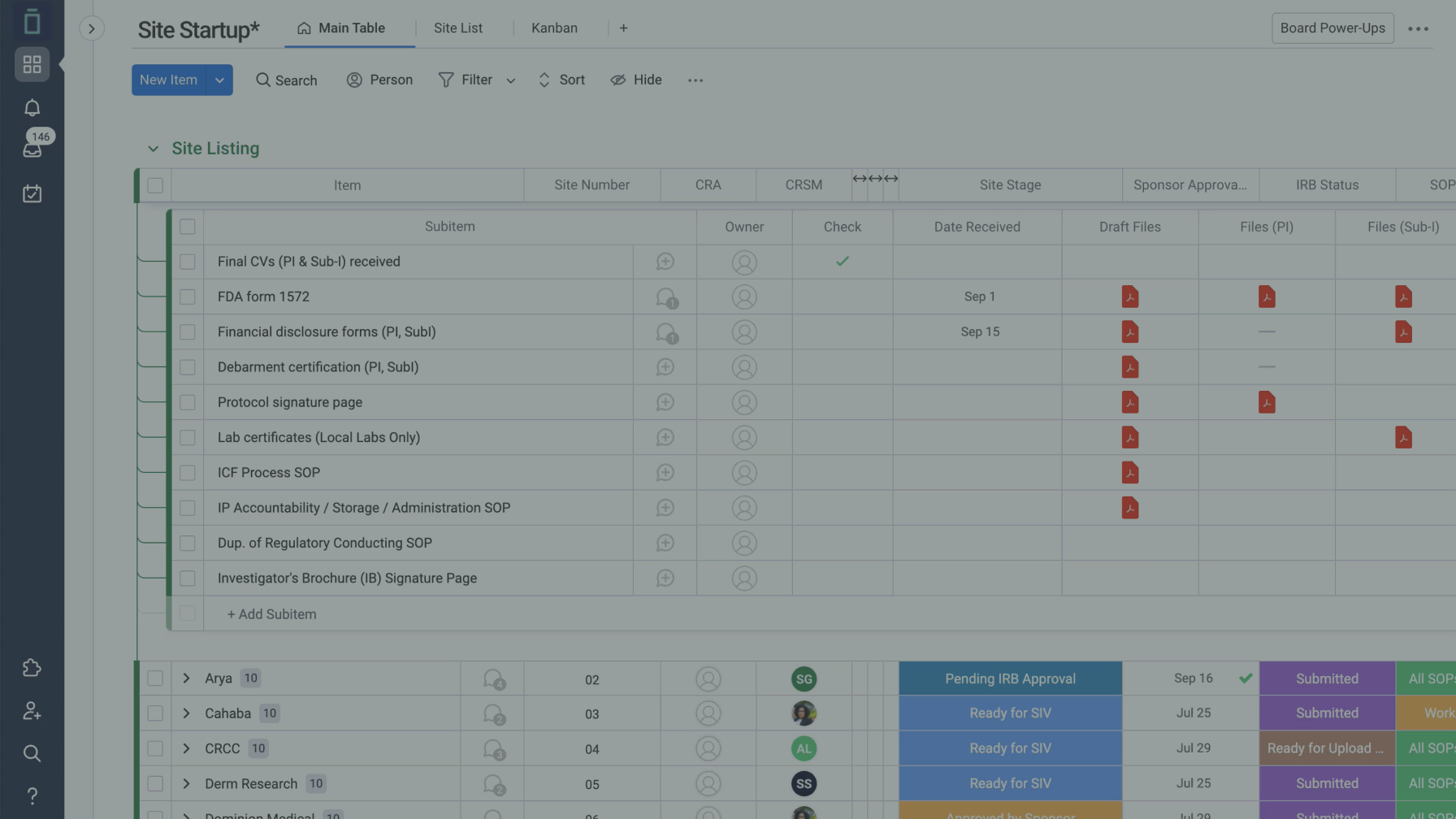
Vial is a next generation Neurology CRO that promises a faster, more accurate, and more efficient clinical trial experience. Its technology platform powers Clinical trials end-to-end and includes an advanced Electronic Source and robust CRA tooling. Base in San Francisco, Vial has over 125 employees and has conducted more than 750 clinical trials. Vial recently expand its Scientific Advisory Board, including Dr. Arvind Chaudhry, to include expertise in Oncology Clinical Trials.
Vial’s mission is to transform the clinical trial industry
The company’s streamline Clinical trials management systems eSource, ePRO, and EDC provide a single solution that streamlines the entire clinical trial process. The Vial team has also create a centralized pre-screening call center to streamline patient recruitment.
Vial’s unique recruiting platform offers clinical trial sponsors faster and more cost-effective Clinical trials. The company’s marketing team, with experience in marketing for $10 billion+ consumer companies, has built a world-class patient recruitment engine. Through its pre-screening call center and EMR filtering capabilities, Vial engages patients and ensures the Clinical trial reaches its enrollment targets.
Vial CRO is a true partner for biotech companies and strives to deliver faster and better clinical trials. With a dedicate recruitment team, a centralized pre-screen team, and a web-base enrollment playbook, Vial offers its clients a customize approach to enrollment that maximizes the success of each Clinical trial. Additionally, the Vial Clinical Network includes more than 35 dermatology clinics in the U.S., which will provide access to a diverse and accurate data set.
Dr. Roberta Brinton joins the Vial Central Nervous System Advisory Board. A leading urological oncologist, Dr. Sonpavde has led transnational projects and developed prognostic endpoints for bladder cancer. She has hold positions at the Dana-Farber Cancer Institute and the University of Massachusetts Medical School. She is also a member of the Southwest Oncology Group and the Blader Cancer Task Force.

Neurological Clinical Research Institute (NCRI)
The Neurological Clinical Research Institute (NCRI) is a nonprofit organization that supports the development of new drugs and therapeutics for neurological disorders. They also support research into the neural mechanisms that underlie neurocognitive disorders. Their goal is to improve clinical trials by supporting scientific collaboration and building the infrastructure necessary to accelerate clinical trials. In addition, they also encourage scientific innovation through funding, training, and education.
The Neurological Clinical Research Institute (NCRI) was found by Drs
Merit Cudkowicz and Steven Greenberg to speed the development of novel treatments for neurological disorders. The institute also is dedicate to developing and training future international leaders in clinical research. Also, They offer graduate and post-doctoral training that provides hands-on, practical instruction in clinical trials. Training includes research design, electronic data capture, and overall principal investigator responsibilities.
The Neurological Clinical Research Institute (NCRI) is compose of various divisions. One division is dedicate to the design and execution of clinical trials for neurological disorders. The other division focuses on bioinformatics and computational methods to analyze and interpret large datasets. These divisions work together to improve the diagnosis of rare conditions, improve therapies, and develop better diagnostics.
The Neurological Clinical Research Institute (NCRI) has a long track record of conducting clinical trials. In collaboration with the Mass General Bio statistics Center, the institute has completed large-scale multi-center Clinical trials for neurological disorders, including ALS. The institute is also the Clinical Coordinating Center of the NEALS Network and the Parkinson’s Study Group.
Read also: What does CRO`s stand for in Clinical research?
QPS
QPS is a Delaware-base CRO with expertise in drug development in the field of neurology. Recently, the company acquire a controlling stake in JSW, one of the leading drug development companies in Austria. Both companies focus on studies involving disorders of the central nervous system. The acquisition gives QPS a deeper expertise in Phase II-IV drug development and expands its footprint in Central and Eastern Europe.
The growing prevalence of neurological diseases and research on these disorders is driving the market for neurology CROs. According to the Parkinson’s Foundation, 1.2 million people will suffer from Parkinson’s disease by 2030. This surge in neurology-related clinical trials will positively impact the neurology CRO market.
QPS Neuropharmacology brings together in vitro and in vivo models with behavioral, biochemical, and histological methods. Using knowledge from over two thousand completed projects, QPS Neuropharmacology delivers the highest quality of results. The company focuses on neurodegeneration models, animal studies, and in-vitro studies.
QPS was find in 1995 by Ben Ross. Today, it employs over 1170 professionals at its nine facilities. It is the recipient of several awards, including the Delaware Bioscience Association’s 2013 Company of the Year award and the Finance Monthly’s CEO of the Year Award. Ben Ross is a former biotech entrepreneur and co-found Theracore Pharmaceuticals, Inc., and Avid Radiopharmaceuticals, Inc., both of which were acquire by Eli Lilly in late 2010.
Cognitive Clinical Trials (CCT)
Cognitive Clinical Trials (CCT) for Neurology CRO is a clinical research organization focused on delivering exceptional quality data. The organization has a team of dedicated professionals who deliver expert support to all study subjects. In addition, Cognitive Clinical Trials (CCT) has developed a hybrid environment that combines traditional clinical research with current technologies. This innovative model has the potential to improve patient access to and retention in clinical trials.
Neurology CRO clinical trials include several phases. These stages include pre and post-interventional assessments. These studies are designed to identify potential new treatments or interventions. They are conducted in a controlled environment and participation is completely voluntary. These Clinical trials are intended to develop new medications or surgical procedures that can benefit patients with neurologic conditions.
CNS CRO and New Partnerships
CNS CRO is one of the largest regional clinical research organizations. The company specialises in early phase non-oncology clinical trials in the region. The company is also internationally recognized as a leading full-service CRO with a focus on global project delivery across all phases.
The CNS-CRO works with a broad range of clients in the development of novel therapeutics. Their expertise ranges from establishing new drug platforms to conducting phase 1 and phase 3 clinical trials. They provide data from diverse sources, including behavioral tests and in vitro and in CNS electrophysiology.
The CRO’s technical expertise is essential to the success of CNS-related clinical trials. The CRO should have deep knowledge of the disease or disorder being studied and the specific conditions that can arise during the study. The CRO should also have a strong working relationship with the IRB and understand the regulatory requirements of the CNS study. This will reduce the risk of protocol amendments and allow for a quicker study start-up.
To improve the ability of the CNS-CRO to test new treatments in neurological disorders, the company has recently signed a licensing agreement with the University of Prince Edward Island (UPEI). The CNS-CRO’s licensing agreement with the UPEI will allow the company to use more clinically relevant animal models for their studies. This new partnership will help the CNS-CRO differentiate itself from existing CRO service providers.





